GENERAL CONSIDERATIONS AND LIMITATIONS OF THE PROPOSED POST-INTRODUCTION OBSERVATION:
We feel that post-introduction observation of new technologies is a strategy which, in great measure, complements procedures implemented in Spain to regulate the incorporation and introduction of new technologies. Although it could be claimed that there is a certain degree of overlap with other activities undertaken during the post-marketing stage of new technologies, we understand that post-introduction observation is a clearly differentiated strategy, not only on account of its different approach, but also because it affords previously unavailable information that could be crucial for ensuring the equity and quality of health services.
To date, follow-up systems implemented after the introduction of new technologies at an international level have fundamentally been designed to assess potentially relevant technologies, for which there is no scientific evidence to determine their safety, efficacy or effectiveness, with the aim of deciding on their definitive funding/approval (monitorised use/monitoring) or evaluating whether the manufacturers' basic requirements as to safety and effectiveness are being complied with. Post-introduction observation of new technologies is a strategy designed for technologies where, despite an apparent absence of problems of effectiveness and/or safety, their characteristics (i.e., they are applied to large population groups, to conditions with high mortality/morbidity, to highly sensitive group, and/or are very innovative, costly, etc.) may nevertheless render it vital for their application and performance to be recorded under real conditions of use, in order to guarantee accessibility and adequacy of use, and detect possible problems of effectiveness and/or safety in the short/medium term. Possessing reliable and timely information is essential for being able to remedy aspects that may influence the quality of the services performed or, on the contrary, for fostering the use of technologies under given conditions (subgroups of patients, modalities of use, etc.).
Given that the present proposal for post-introduction observation is based on epidemiological principles solid numerators and denominators can be obtained. Moreover, the fact that it is conceived as an active data-collection system means that the great shortcoming of surveillance systems, i.e., low reporting rate and possible information bias, can be remedied.
Table 7 shows the main differences between the follow up systems established in Spain, which might be common to many other developed countries.
| Table 7. Principal health-technology follow up systems established to date in Spain. | ||||
|---|---|---|---|---|
| Principal health-technology follow up systems established to date in Spain. | ||||
| Yellow card | Monitored use | Medical device vigilance systems | Post-introduction observation | |
| Target technology | Medications | Techniques, technologies and procedures (except medications) | Medical devices | Any health technology (except medications). |
| Effectiveness of technology | Known | Not totally known | Known | Known |
| Purpose | To ascertain low-frequency, severe adverse effects | To ascertain effectiveness and safety | To ascertain any dysfunction or deterioration in the characteristics or operation of the medical device which may have caused or causes the death or deterioration of the patient's health. | To ascertain diffusion, acceptability, adequacy of use and detect problems of effectiveness, adverse effects and costs, particularly those that arise in the short-/medium-term. |
| Participants | Physicians and pharmacists | Physicians | Manufacturers and end-users (in most cases) | Health professionals, patients and industry |
| Selection of participants | None | Ministry of Health & Consumer Affairs | None | Centres that may incorporate the technology. |
| Level of implementation | National | National | National | National |
Post-introduction observation of new technologies is clearly differentiated from monitoring because it is formulated as a structured strategy to identify, collect and analyze relevant information that is collected routinely during clinical practice. The current proposal, in contrast to monitoring, requires a minimum investment of time from clinicians and provides them with direct information of the real problems concerning the application of technologies in real practice. However, as in other follow up systems, clinician's must be made aware of the value of the information obtained to guarantee their collaboration. All health professionals involved in the utilization of the technology must be implicated. Similarly, to set up these systems time and resources are needed and these can only be obtained with adequate financing.
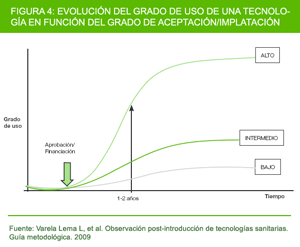
For practical reasons, it is proposed that post-introduction observation should cover the initial stages of implementation and continue for a minimum period of 1 year after the new technology is approved. An adequate period of time must be allowed to elapse so as to detect adverse effects that appear in the medium term and identify any possible problems of implementation and/or acceptability. It is foreseeable that the use of new technologies by health professionals will evolve in line with the innovation diffusion model proposed by Beals et al (26). According to this model, new technologies will spread in the form of an S-shaped curve, with diffusion passing through 5 differentiated stages, namely: 1) from first knowledge of an innovation; to 2) forming an attitude toward the innovation; to 3) a decision to adopt or reject) to 4) implementation of the new idea; and to 5) confirmation of this decision. Based on this theory, we feel that 1 year could be sufficient for detecting possible variations with respect to what was originally foreseen. This period of time will likewise be enough for detecting adverse effects that may arise in the short-/medium-term, though this is preliminary information, and thus it might well be of interest in the majority of cases for this follow-up period to be extended so as to detect the long-term adverse effects as well. However, we see this as being somewhat unfeasible, in view of the great cost that it -together with the introduction of other alternative technologies- would entail, and it is therefore only recommended in specific cases where technologies are applied to a small number of patients or analysis by subgroup must be performed
Figure 4 depicts the way in which the use of technology would evolve where the degree of acceptance/implementation was: 1) high; 2) intermediate; and 3) low.
The current methodological guideline outlines the specific procedures and strategies for planning, implementing and assessing the use of new technologies. It is a consensus-based document that was drawn up by a working group made up of experts in health technology assessment, with the participation of managers/administrators, clinicians and patients, who are the main parties involved in the planning and use of new technologies. This renders it quality and favours its acceptance. The main aim of publishing the guideline in an abridged format, with the results shown in schematic tables, is to favour its use. There is evidence to show that presentation in this more practical format is a factor that greatly facilitates its use in health policies (27).
This is the first document to be published on the topic and it is thought that it could serve as a point of reference for any institution/body, national and international, which may be involved in planning and/or implementing post-introduction observation activities. Similarly, scientific societies could also make use of this document for the promotion of new technologies, where clearly beneficial results are obtained.
However, despite the fact that the methodology might be extrapolable to other countries with similar health systems, it must be stressed that certain adaptations might be required since it is fundamentally based on the opinion of experts and experience obtained in a specific context (Galicia). In the following paragraphs the main considerations for each of the 3 sections of the guidelines are discussed.
In this part