POST-INTRODUCTION OBSERVATION OF NEW TECHNOLOGIES IN THE CONTEXT OF HEALTH TECHNOLOGY ASSESSMENT
The great technological advances of the last 50 years have posed an important challenge to most health care systems around the world. Ensuring innovative quality care, which adjusts to the demands and expectations of end-users, managing budgets allocated to health, and safeguarding the basic principles of equity and accessibility is a very complicated task. Health technology assessment (HTA) is a multidisciplinary field of research which was created in the 1970s for the purpose of providing objective information to medical decision-makers and politicians, to support decision making about the introduction of new technologies (1). HTA was fundamentally developed to review aspects of safety, efficacy, cost and cost-effectiveness of new health technologies, and to assess the clinical, social, financial, organisational and ethical implications, both direct and indirect, stemming from their adoption, along with their desired and undesired effects.
Since its origins, HTA has been steadily evolving and extending its activities so as to adapt to the exponential growth in technological innovations and the important changes in the priorities of a society that is increasingly aware of its state of health and ever more demanding when it comes to therapeutic options. At the outset, HTA basically consisted of a comprehensive evaluation of the scientific evidence in order to provide support for decision-making with respect to the introduction of new technologies, with health technology being defined as, "the equipment, devices and drugs and the medical and surgical procedures used in prevention, diagnosis, treatment and rehabilitation of disease, as well as the organisational and support systems used in the delivery of health care" (2).In the 1990s, the need arose for new and emerging health technologies to be detected and assessed prior to their marketing and/or diffusion, in order to provide managers and administrators with useful and timely information on technologies eligible for incorporation into clinical practice, and to facilitate the planning of activities, budgets and resources. Currently, a number of countries, including Spain, have implemented early warning systems for the identification and assessment of new and emerging health technologies [3-5].
In the last decade, in view of the great pressures to incorporate insufficiently assessed technological innovations, a number of initiatives have been established to monitor new and emerging health technologies before they are adopted by the health care system. Such monitoring is seen as a measure that allows for the conditional introduction of new technologies subject to collection of additional data. They are fundamentally designed to provide information on technologies which have potential to produce major improvements in health or quality of life, but there are doubts as to their safety and/or effectiveness (6).The need to monitor new technologies is included as a recommendation in many HTA reports carried out by Spanish HTA agencies (avalia-t, AETS, Osteba, etc) and by other agencies like CEDIT ((Comite d'Evaluation et de Diffusion des Innovations Technologiques).or ANZHSN ( Australian and New Zealand Horizon Scanning Network. At the level of the Spanish National Health System (NHS), conditional monitoring of new technologies is known as "monitorised use" (uso tutelado) and is considered within the legislation as a mechanism to generate and collect data in order to establish the degree of safety, efficacy, effectiveness or efficiency of a given technique, technology or procedure to inform decision making regarding its approval as a covered health benefit (RD 1030/2006) (6). In the Autonomous Region of Galicia (7) and País Vasco (8) the specific legislation establishes that, based on the available scientific evidence, some technologies might be proposed for conditional monitoring and be subject to specific centres, indications, protocols and assessment criteria. [7,8].
Despite the fact that HTA has become a tool of great value for making decisions about the approval/funding of new technologies, many uncertainties are frequently associated with the introduction of new technologies that can only be assessed after their definitive approval and adoption in clinical practice (9). Preliminary scientific information is insufficient to assess the real impact that the new technology is going to have on the population or to assure that anticipated expectations are accomplished.
When a technology is approved, there is a possibility that diffusion and adoption does not take place as expected, owing to financial adversities, to the existence of organisational, structural or technical constraints or to the rejection by clinicians or patients. Similarly, it is likely that diffusion at 1st-level hospitals may not be the same as at 2nd- or 3rd-level hospitals, and this could give rise to important differences of accessibility to technological innovations for the different health areas or regions. These facts, linked to the different degrees of willingness of health professionals and/or institutions to use innovative technologies, can result in important variations in their use [10,11]. In some cases, institutions' competence and their desire to try new technologies can lead to over-use of such technologies, with the ensuing increase in costs and resources but without any proven benefit for the patient or with even greater adverse effects. In other cases, resistance to the use of new technologies could greatly influence the quality of the services offered.
The medical practice is based on the acquisition of knowledge and skills that should ensure professional aptitude and quality. Nevertheless, when new technologies are involved, there are frequently many doubts as to proper technology use, the selection of patients and as to the applicability of research results to daily clinical practice (9). It is foreseeable that when clinical indications are based on subjective criteria (e.g., seriously ill patients, not eligible for other treatments, etc.), there may be great heterogeneity in the selection of patients eligible for using the new technology is concerned. Deciding which patients are to be diagnosed/treated with the new technology may greatly depend on the occupational and social context (organisational structure, available resources, degree of specialisation, learning curve, values, internal policies, citizen pressures, etc.), and one consequence of this is the appearance of important variations in medical practice (12). Variations in medical practice are especially relevant when technologies use can clearly impact on patients' mortality, morbidity and/or quality of life. Radiofrequency ablation, indicated for hepatic metastasis of colorectal cancer, is a good example of a technology prone to displaying wide variability, with important repercussions on population health (13).
On applying the technology under normal conditions of use, there is also the possibility that problems of ineffectiveness or side-effects unforeseen in pre-marketing studies might appear (14).Pre-marketing studies are conducted by groups of experts on small groups of patients and in modified conditions. It may happen that severe side-effects are not detected because these only appear in a small number of patients, yet the possibility also exists that problems of ineffectiveness and/or safety may arise due to clinicians' limited experience (learning curve). In techniques that require a high degree of specialisation, such as the case of hemiliver transplants from living donors, very important differences have been observed in morbidity and mortality depending on the volume of procedures performed (13). Side-effects or problems of effectiveness may also arise as a result of the new technology beingused on highly sensitive groups (children, the elderly, patients with other co-morbidities), or among the health personnel responsible for its application. Likewise, inappropriate use or the combination of new technologies with other procedures or devices could also give rise to interactions and side-effects unforeseen in the literature. Such was the case of the combination of cholecystectomy with laparoscopy; techniques that separately displayed a high degree of safety but when combined gave rise to a great increase in the rate of biliary lesions. Investigating and quantifying adverse effects and/or problems of effectiveness is relevant for taking appropriate measures as soon as possible and ensure the health of the population (13).
The different post-introduction follow-up systems regulated world-wide are basically surveillance systems developed to report on the serious adverse effects of medical devices and suffer from an important degree of underreporting [15,16].Problems associated with the overuse of new technologies in dubious/non authorized indications, lack of effectiveness, incompatibility with other treatments or complications that may appear only when the technologies are used in specific groups are frequently not identified by the available follow-up systems. Neither are structural, organizational or economic problems that can lead to an underuse of the technology.
The investigation of problems after the approval of technologies has basically relied upon the implementation of voluntary registries set up by different associations or independent research studies carried out by the health services. These studies, even though of great value, present the limitation that they are frequently not developed following a structured methodology and are undertaken in may cases only when clinicians become aware that there might be problems with the technologies. Commonly these investigations cover only areas of high clinical interest (cardiovascular diseases, cancer, etc) and are basically aimed at assessing specific aspects of effectiveness, safety or variability of use and do not offer information on other aspects that may be relevant to guarantee the quality of the health care [17-19].
The development of a systematic, prioritised process for collecting, analysing, interpreting and disseminating information on the utilization of new technologies, once these have been introduced into daily clinical practice is necessary for ensuring the quality of the health-care. It would allow for the identification and assessment of problems concerning implementation, accessibility, acceptability and adequacy of use, and would provide reliable and comprehensive information on the real costs and use of resources that are derived from its application. Since it integrates different sets of data generated at local levels, it allows for the detection and assessment of relatively rare adverse effects or problems of effectiveness that are only observable with large sample sizes or in specific populations. Having large population groups for specific indications would also make it possible to identify specific subgroups with a high risk of suffering from adverse effects that would otherwise go undetected in preliminary studies. Equally, it would enable assessment of other aspects which might influence effectiveness and safety (structural, organisational, training-related, etc.). The main goals of a post-introduction system for observation of new technologies are outlined in table 1.
| Table 1. Main objectives of a post-introduction observation system of new technologies. | |
|---|---|
| OBJECTIVES | |
| General objectives | Specific objectives |
| 1. To assess the diffusion of the new technology |
|
| 2. To assess accessibility within the Health System |
|
| 3. To assess adequacy of use |
|
| 4. To verify effectiveness |
|
| 5. To verify safety |
|
| 6. To verify economic impact |
|
At present, there is no systematic, prioritised process in Spain, or in any other country in the region, for compiling information on new technologies once these have been approved and widely diffused throughout the health system.
Like other governmental associations/organisations, including the Australian Government Productivity Commission and the European Observatory on Health Systems and Policies [1,20,21],we feel that post-introduction assessment of new technologies is essential to inform decision-makers about the real implications deriving from the introduction of new technologies, to furnish relevant information on problems of implementation, accessibility, acceptability, adequacy of use and economic impact, and to identify specific problems of effectiveness and/or safety.
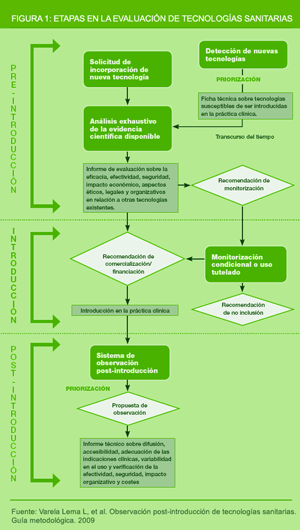
In line with the relevance of continuing HTA in the post-introduction stage, we propose that HTA must integrate 3 stages (pre-introduction, introduction and post-introduction) and comprise the following steps:
- Early identification and assessment of new and emerging health technologies.
- Comprehensive assessment of scientific evidence to provide support for decision-making about the approval of new technologies.
- Conditional monitoring of new, insufficiently assessed technologies in order to establish the degree of safety, efficacy, effectiveness or efficiency of a given technique, technology or procedure and inform decision making regarding its approval as a covered health benefit.
- Observation of new health technologies once they have been approved/financed by the health care system and are used in daily clinical practice.