LEGAL FRAMEWOK FOR THE INTRODUCTION OF TECHNOLOGIES IN SPAIN
At a national level, health-technology assessment and monitoring are envisaged by law as indispensable procedures for decision-making with regard to the incorporation of new techniques, technologies or procedures into the National Health System's common service portfolio, but this does not apply to post-introduction observation. The latest Ministry of Health & Consumer Affairs Order 3422/2007 of 21 December puts forward that (24),when the scientific evidence available is deemed insufficient for decision making regarding the generalised financing of new techniques, technologies or procedures in the NHS, a proposal for an assessment study must be formulated via monitored use or some equivalent mechanism.
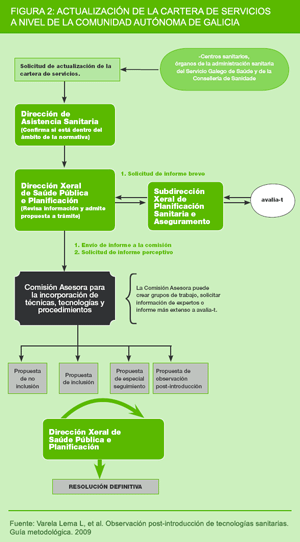
Within Spain, the Autonomous Region of Galicia has drawn up a specific statutory regulation to establish the procedure for incorporating techniques, technologies, procedures or other means of delivering health-care which are included in the NHS portfolio, into the Galician Health Service (Official Galician Gazette - Diario Oficial de Galicia/DOGA; order of 7 July 2003) (7). Article 5.2 provides that some technologies may be deemed eligible for special follow-up (monitored use), and prior to their definitive incorporation be conditioned to specific centres, indications, performance and assessment criteria. Although it is not envisaged under current legislation, certain health technologies are subject to a process of post-introduction observation, aimed at conducting a follow-up of their use after they have been introduced into the service portfolio. Unlike special follow-up, the use of this technology is not restricted to special centres because the intention is to evaluate their use under real conditions. Figure 3 depicts the procedure for incorporation of new technologies in Galicia:
>